β-Spectroscopy Experiments
β-Spectroscopy introduces the student into the field of special relativity and weak interactions of radioactive decays.
After radioactivity discovery, E. Rutherford separated radioactive emissions into two types: alpha (α) and beta (β–) radiations, based on matter penetration and ability to cause ionization. The β– particles have higher penetration and lower specific ionization than alpha particles. Beta particles are electrons (β–) or positrons (β+), resulting by nucleon decays in unstable nuclei. Since beta decay is a three body process, the β energy spectrum is continuum.
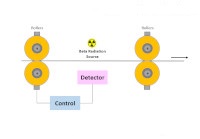
The β-Spectroscopy experiments are performed using the Educational Beta and Premium Kits.