| Difficult | Execution Time | Data Analysis | Radioactive Sources |
|---|---|---|---|
| Yes | No |
Hardware setup
This experiment guide is referred to the SP5630EN educational kit.
Equipment: SP5630EN – Environmental kit
| Model | i-Spector – S2570B | Samples |
|---|---|---|
| Description | Intelligent Silicon Photomultiplier Tube | Empty Beaker & Test Sample, Fertilizer and Rock, Canisters of Activated Carbon, Calibration Crystal |
Purpose of the experiment
Record the energy spectrum of the Test sample and identify the peaks, after the energy calibration, by knowing the decay chain of Thorium and Uranium.
Fundamentals
Naturally occurring radiative material (NORM) is material found in the environment that contains radioactive elements of natural origin (uranium, thorium, and potassium). NORM is often found in its natural state in rocks or sand but it can also be present in consumer products, including common building products (like brick and cement blocks), granite counter tops, glazed tiles, phosphate fertilizers, and tobacco products. Moreover, there are some of the materials and products sitting around your house could be emitting low levels of radiation. In the past, radioactive materials were employed by humans in objects of common use, like ceramics dishes (once uranium oxides were used to create a bright red-orange dinnerware), drinking glasses (glassmakers widely used uranium to color glasses a transparent yellow or yellow-green), clocks (glow in the dark with radium in the paint), camping lanterns (white light due to thorium), etc.
The test sample available in the kit contains old objects containing radioactive elements. The user can acquire the energy spectrum and try to recognize the source by knowing the decay products of Thorium and Uranium.
Carrying out the experiment

Experimental setup block diagram
Put the i-Spector digital into the base and place the Test sample box into the place-holder. Power on the i-Spector and connect the Ethernet cable. Wait until the temperature is stable from the web interface (it can take half an hour from power on).
Check the waveform, modify the threshold and gate width, if needed, then start the measurement of the energy spectrum.
Take few minutes of acquisition according to the sample activity
NOTE: in case of background measurement, settings and acquisition time must be the same. Lead blocks could help in distinguish clearer peaks but they must be used if just employed in background measurement only.
Results
The background subtraction can be made with the background spectrum. After that, it is possible to recognize peaks and identify the source, either Thorium, Uranium, or both, as shown in the picture below. The user can identify which source is contained in the Test sample by knowing the product decay of the two elements.
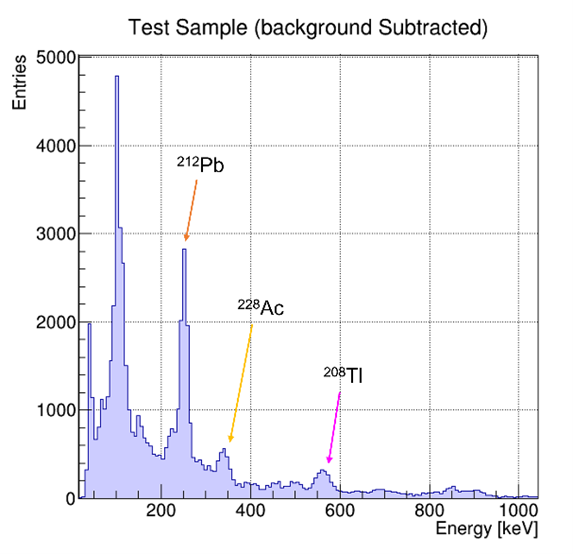
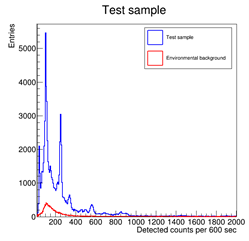
Test sample: total contribution and background on the left; background subtracted on the right. The visible peaks are highlighted in the spectrum



